Advances in technology and decreased testing costs have led to a rise in the number of genes associated with inherited cancer risk for which testing is clinically available. At the same time, national practice guidelines have been revised to reflect the changes in expanded test offerings.
One disease site that recently underwent significant guideline changes by the National Comprehensive Cancer Network (NCCN) is colorectal cancer (CRC). NCCN updated its Genetic/Familial High-Risk Assessment: Colorectal guidelines this year to include several genes newly associated with increased CRC risk. In addition, management guidelines for more well-described syndromes, such as Lynch syndrome (LS), were also modified.
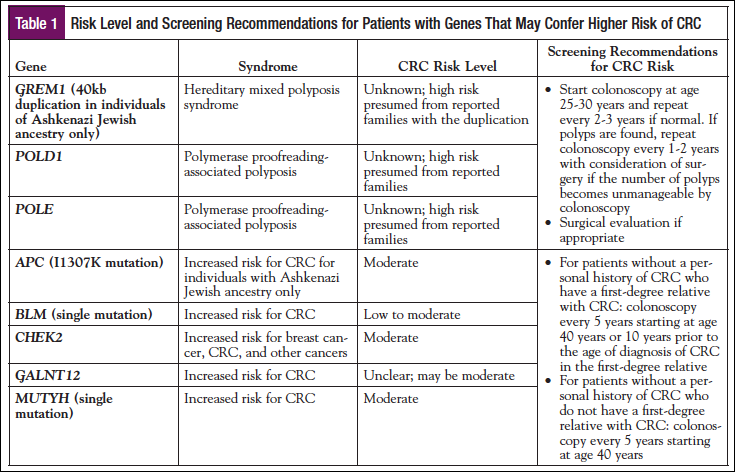
Summary of NCCN Modifications to CRC Gene Guidelines
GREM1—A 40kb duplication upstream of GREM1 in individuals of Ashkenazi Jewish ancestry is associated with hereditary mixed polyposis syndrome. Currently, the lifetime CRC risk is unknown but assumed to be high. Colonoscopy should begin between ages 25 and 30 years and be repeated every 2 to 3 years if normal. If polyps are found, colonoscopy should be repeated every 1 to 2 years and removal of the colon considered if polyp burden becomes unmanageable.
POLD1 and POLE—Mutations in POLD1 and POLE are associated with polymerase proofreading-associated polyposis. Currently, the lifetime CRC risk is unknown but is assumed to be high. The phenotype appears to overlap with LS (typically 0 or few colonic polyps) and familial adenomatous polyposis (FAP; hundreds of colonic polyps). Colonoscopy should begin between ages 25 and 30 years and be repeated every 2 to 3 years if normal. If polyps are found, colonoscopy should be repeated every 1 to 2 years and removal of the colon considered if polyp burden becomes unmanageable.
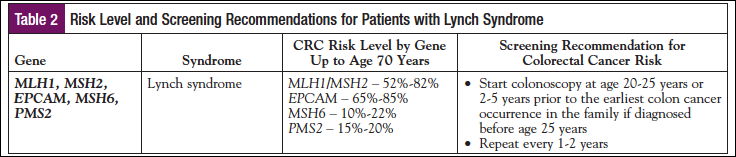
MLH1, MSH2, MSH6, PMS2, EPCAM—Mutations in MLH1, MSH2, MSH6, PMS2, and EPCAM are associated with LS. In addition to colon cancer, LS places individuals at increased risk for a variety of other cancers. The lifetime CRC risk to age 70 years varies depending on the gene harboring the mutation and ranges from approximately 10% to 85%. Guidelines were recently modified to recommend colonoscopy at the same age and same interval, regardless of which gene has the mutation. Colonoscopy should begin between ages 20 and 25 years or 2 to 5 years prior to the earliest CRC in the family if that cancer was diagnosed at younger than age 25 years. Colonoscopy should be repeated every 1 to 2 years.
NCCN Updates for Other Genes Conferring Increased Risk of CRC
Recent updates suggest that individuals with the APC I1307K variant, single mutation carriers in BLM or MUTYH, or mutation carriers in CHEK2 or GALNT12 should undergo colonoscopy at a younger age and at increased intervals. For carriers with a first-degree relative (parent, sibling, child) with CRC, colonoscopy should begin at age 40 years, or 10 years prior to the earliest CRC diagnosis, and be repeated at least every 5 years. For patients without a first-degree relative with CRC, colonoscopy should begin at age 40 years and be repeated at least every 5 years.
APC I1307K variant—Mutations in the APC gene are typically associated with attenuated familial adenomatous polyposis (AFAP) or FAP. Both syndromes are characterized by a high polyp burden, typically greater than 10 and with FAP often greater than 100. However, the I1307K variant is unique in that its presentation is not classic, and polyps are not always seen. The variant confers an increased risk for CRC and is typically seen in individuals of Ashkenazi Jewish descent. For carriers with a first-degree relative with CRC, colonoscopy should begin at age 40 years, or 10 years prior to the earliest CRC diagnosis, and be repeated at least every 5 years. For patients without a first-degree relative with CRC, colonoscopy should begin at age 40 years and be repeated at least every 5 years.
BLM (single carrier/1 mutation)—Individuals who carry a mutation in both of their BLM genes have Bloom syndrome, which is characterized by short stature, increased sensitivity to light, immune deficiency, and an increased risk of cancer. Data are beginning to suggest that individuals carrying 1 mutation have an increased risk for CRC.
CHEK2—Mutations in CHEK2 are associated with an increased risk for CRC and several other cancers.
GALNT12—Mutations in GALNT12 appear to be associated with an increased risk for CRC, although it is unclear if the risk is low or moderate.
MUTYH (single carrier/1 mutation)—Individuals who carry a mutation in both of their MUTYH genes have MUTYH-associated polyposis syndrome and a high risk of developing CRC. Recent data are beginning to suggest that individuals carrying a mutation in only 1 of their MUTYH genes may also have an increased risk of developing CRC, although not as high as in biallelic carriers.
See Table 1 for a summary of NCCN modifications to CRC gene guidelines and NCCN updates for other genes conferring increased risk of CRC.
NCCN Updates for Genes Associated with LS
In addition, the updated guidelines combined colorectal surveillance recommendations for individuals who carry a mutation in any of the 5 genes associated with LS. Previously, the colorectal screening recommendations were different for carriers of MLH1, MSH2, or EPCAM mutations (which have the highest lifetime colon cancer risks) versus carriers of MSH6 and PMS2 mutations (which have lower lifetime colon cancer risks). It is now recommended that colon cancer surveillance begin at the same age and at the same interval, regardless of the gene mutation. This is an important update for individuals with LS to share with their at-risk family members, as this adjustment may help to inform the age at which relatives may consider predictive testing for a known mutation in the family. See Table 2 for a summary of the current NCCN guidelines for CRC surveillance for patients with LS.
Conclusion
It is important to keep in mind that as our knowledge about many of the inherited genes associated with cancer risk continues to evolve, risk estimates and management guidelines will likely continue to undergo updates. There are many genes included on clinically available tests that do not yet have precise lifetime cancer risk estimates or corresponding practice guidelines, which makes these test results challenging for healthcare providers. Staying abreast of any updates to cancer risk estimates and guidelines will enable clinicians to offer additional genetic testing options that may be appropriate for patients who have previously undergone genetic testing, and to modify their medical management of patients who have previously tested positive.
Reference
National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines). Genetic/Familial High-Risk Assessment: Colorectal. Version 1.2016. www.nccn.org/professionals/physician_gls/pdf/genetics_colon.pdf. Accessed September 21, 2016.