Background: It is strongly recommended that individuals ending treatment for cancer have a “survivorship plan,” and new standards require survivorship planning for accreditation. However, a comprehensive plan is often neglected.
Objective: To present the development and field test results of a web-based, breast cancer survivorship care planning system.
Methods: The Polaris Oncology Survivorship Transition (POST) blends input from the electronic health record (EHR), oncology care providers (OCPs), and patients to create a survivorship care plan (SCP). The content of the POST program was created with the assistance of end-user input (patients, oncologists, and primary care providers [PCPs]) and the full program was piloted on women ending treatment for breast cancer. This paper presents the pilot study that field-tested the POST in a clinical setting. Patients were recruited from outpatient care clinics and chemotherapy units in a comprehensive care center. The study included 25 women ending treatment for breast cancer in the past year, 4 OCPs, and PCPs. Patients received the POST computerized assessment and a tailored SCP.
Results: The POST assists providers in crafting efficient and comprehensive SCPs and was rated highly satisfactory by all end-users.
Discussion: The POST program can be used as a cancer survivorship planning program to assist OCPs in care planning for their patients ending treatment for breast cancer.
Conclusion: This study provides support for incorporating computerized SCP programs into clinical practice. Use of the POST in clinical practice has the potential to improve survivorship planning.
Within the United States, the estimated number of cancer survivors will increase from 14.5 million to approximately 19 million by 2024.1 The National Coalition for Cancer Survivorship distinguishes between 2 phases of survivorship: 1) intermediate/short-term (posttreatment), and 2) long-term survivorship. In 2015, the American College of Surgeons (ACS)2 implemented accreditation expectations that include a written or electronic survivorship care plan that is 1) prepared by the OCP; 2) given to patients at treatment completion; and 3) includes a record of care received, important disease characteristics, and a follow-up plan incorporating evidence-based standard of care.
Survivorship care planning has been a highly debated topic in cancer care. Whereas some oncology care experts argue that survivorship planning has the potential to improve quality of life, decrease morbidity and mortality related to future cancers, and enhance patient access to resources to manage the physical, emotional, and social sequelae of cancer treatment,3-6 others argue that there is a lack of evidence that survivorship care planning actually impacts patient outcomes.7 One of the reasons for the debate is that there is indeed a lack of methodologically rigorous studies examining the outcomes of care planning, which is likely due to care planning being time-consuming and, thus, not done consistently by OCPs.8 One exception is a study by Grunfeld and colleagues,9 which is one of the only randomized controlled trials that has tested outcomes related to survivorship planning and has received much attention because of its null findings. Primary critiques include: a lack of tailored survivorship planning; patients out of cancer treatment for an extended time; extrapolating findings from the Canadian healthcare system to the US system; time of follow-up measurement; and use of noncancer-specific distress tools.10,11 Further, Grunfeld’s survivorship planning did not include any assessment of the patient’s status or needs and neglected to integrate care planning into clinical practice.
The purpose of this paper is to present the findings from a phase 1 STTR (small business technology transfer) study, which funded the development of the Polaris Oncology Survivorship Transition (POST) system. The POST is a computerized, web-based survivorship planning program that generates a tailored SCP. The POST system is the first technology-enabled system to produce tailored survivorship plans fully reflecting the Institute of Medicine (IOM) recommendations4 and the ACS 2015 requirements. The POST is innovative as it incorporates information from both the OCP and the patient; features readily available “plug in” for 2-way integration with diverse EHRs; provides dynamic, electronic referrals for specialized support service; and facilitates care coordination between OCPs and PCPs. We also discuss our ongoing phase 2 study, which is examining the POST in a randomized controlled trial to determine its impact on patient outcomes as they move into long-term survivorship.
Method
Participants
Twenty-five patients with breast cancer were enrolled in the field study. Inclusion criteria included being 18 years of age or older, female, having a diagnosis of nonmetastatic breast cancer, and being within 3 visits of ending active treatment or up to 1 year out of ending active treatment for breast cancer. Patients with other types of cancer, men, and patients with communication difficulties were excluded.
Assessments
The POST Patient Assessment was the main outcome measure for participants and a tool for creating the patient-driven portion of the SCP. We considered the assessment a measure for the study as it assesses important outcome variables described below. In addition, patients completed a Preparing for Life as a New Survivor (PLANS) assessment before and after the Patient Assessment, a feedback/satisfaction survey after reviewing their SCP with their OCP, and a follow-up assessment 1 month after enrollment. OCPs and PCPs also completed follow-up assessments.
The POST Assessments
The POST program incorporates 2 data sources to generate an overall SCP : 1) a Provider Questionnaire populated by information from the EHR, which for this study was entered by a research assistant and OCP, and 2) a computerized Patient Assessment (Table 1). The POST SCP is broken down into 2 portions: 1) the provider version of the SCP, and 2) the patient version of the SCP. Both summaries consist of 7 sections: 1) Medical Diagnosis and Treatment Summary, 2) Medical Care Plan, 3) Physical Symptoms and Functioning, 4) Psychological Adjustment or Psychological Distress, 5) Short-term and Long-term Effects of Breast Cancer, 6) Supportive Care, and 7) Cancer-Related Health Behaviors. Sections 1 and 2 of both the provider and patient versions of the SCP are generated from the Provider Questionnaire and are exactly the same in terms of content. Sections 3 through 7, which are further broken down into subsections, are generated from the patient assessment and vary depending on how the patient responds to the assessment. Also, the patient and provider versions of sections 3 through 7 vary in terms of depth of content, as the patient version is significantly more detailed in terms of psychoeducation and resources.
Section 1 of the SCP provides a comprehensive summary of cancer diagnosis and treatment and other notable medical diagnoses. Section 2 summarizes the patient’s plan for the next year, including future scans, tests, and appointments anticipated by the OCP. Sections 3, 4, and 5 include tailored feedback and evidence-based psychoeducation about emotions, relationships, side effects, and lifestyle choices that impact quality of life and health in survivorship. Section 6 is linked to questions regarding social support, spirituality, or financial issues. Finally, section 7 is informed by questions related to diet, exercise, smoking, and alcohol use. This section provides empirically supported information about health behaviors and cancer risks and includes tailored recommendations for behavior change. The “Patient drug information” from uptodate.com was also provided for any current cancer medications taken by the patient. In addition, based on responses from the patient assessment, individuals could request a referral to a psychologist and/or a social worker. For example, individuals with elevated distress were asked if they wanted their information sent confidentially to a psychologist specializing in cancer-related psychosocial issues.
Whereas both the provider version and patient version of the SCP contain information on these 7 areas related to survivorship, as stated above, the provider version is very brief and is focused on simply documenting status and symptoms of the patient. Because the provider version would be saved to the EHR, and potentially sent to the patient’s PCP, we crafted a brief version of the care plan. Hence, we labeled this brief version the provider version or “provider summary.” The provider version is approximately 3 to 5 pages and flags any concerns reported by patients during their assessment (ie, smoking status, high level of depression, sexual problems). (See Appendix A for a Provider Version/Summary SCP example.)
The patient version of the SCP is more detailed, providing information and resources depending on the patient’s responses to the assessments related in sections 3 through 7. Depending on these responses, a patient version of the SCP can range from 25 to 45 pages. This document can be lengthy because if a patient reports concerns about any of the areas assessed, the POST is programmed to provide the patient with printed materials related to the topic as well as regional and local resources that may help them address the reported concerns. For example, if a woman reports that she is concerned about her weight at the end of cancer treatment, the POST will add a section to the patient version of the SCP that provides her with information taken from good resources (eg, American Cancer Society, National Cancer Institute [NCI]) about cancer risk and obesity, as well as information about best ways to manage weight, and will even include some resources in the area for obesity management treatment. We chose to not include an example of a patient version SCP because of the length of the document, but readers are encouraged to contact the authors if they would like to request an example.
The PLANS
The 17-item PLANS scale was used to assess how prepared and knowledgeable women felt regarding surveillance and care as they entered survivorship.12,13 Part 1 uses a 4-point Likert scale (strongly disagree = 1 to strongly agree = 4) to determine patients’ beliefs regarding their role and their providers’ roles during survivorship. Part 2 uses a 10-point Likert scale to measure participants’ confidence in ability to care for themselves as they enter survivorship, with higher scores indicating greater confidence.
Feedback/Satisfaction of POST
The Patient POST Feedback/Satisfaction Survey asked participants about the online assessment process and how they felt about their SCP. Patients were asked how much they agreed or disagreed with 14 items using a 5-point Likert scale (strongly disagree = 1 to strongly agree = 5), open-ended questions, and their ratings for overall satisfaction.
Follow-Up Assessment
The 1-month follow-up survey was adapted from a survey developed by Brothers and colleagues14 to evaluate the clinical utility and impact of survivorship plans and used a 5-point Likert scale (strongly disagree = 1 to strongly agree = 5).
OCP Satisfaction Survey
OCPs evaluated the SCPs by answering how much they agreed or disagreed with statements on a 5-point Likert scale, with higher scores indicating more agreement. They also provided overall satisfaction ratings, feedback about the most useful sections of the SCP, and open-ended comments.
PCP Satisfaction Survey
PCPs were asked about their perception of the SCP’s usefulness for evaluating their patient’s status and future care needs and other ways that the SCP may have been helpful in transitioning care from the oncology team back to the PCP. Similar to the OCP survey, a 5-point Likert scale was used, with higher scores indicating more agreement.
Procedure
Patients
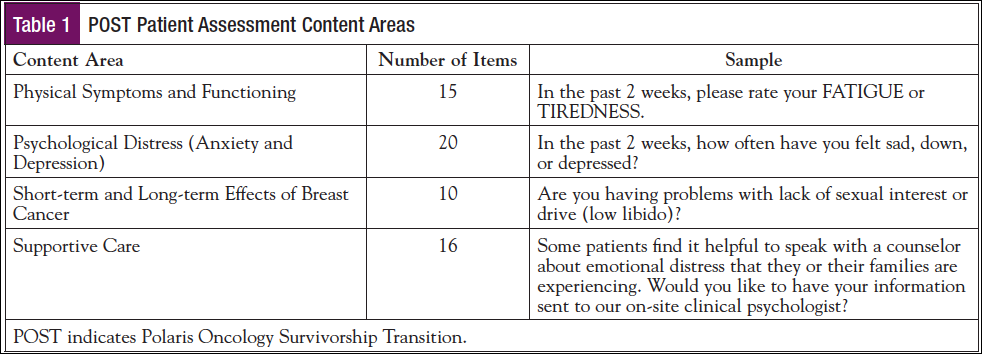
Enrollment procedures are illustrated in Figure 1. Researchers worked with the oncology team to identify potential participants for the field test. All participants went through informed consent procedures and were given the option to send a copy of the provider version of the SCP to their PCP. Participants used an iPad or tablet computer to complete assessments. When the patient portion of the assessment was completed, the OCP completed his or her portion of the POST Provider Questionnaire and checked for inaccuracies. The SCP was then reviewed with the patient and checked for accuracy, and a hard copy was provided to the patient to review at her leisure. After the appointment and SCP review, research staff administered a feedback survey and readministered the PLANS. Any referrals requested for a social worker or clinical psychologist were confirmed with the patient and sent to the appropriate provider. All participants were called 1 month after the initial assessment to complete a follow-up assessment over the phone with a trained research assistant and were remunerated $40.
Only estimates of time were documented for each step taken in survivorship planning for the field test because the POST was still in development and was tested in live clinical situations. Without EHR integration, a research assistant took about 30 to 45 minutes to prepare the Provider Questionnaire, and the OCPs spent about 5 minutes checking and updating the questionnaire after patients completed their assessments. A majority of patients completed their assessments within 10 minutes. It took approximately 10 to 15 minutes to review the SCP with patients at the end of their appointment.
OCPs
OCPs were contacted in person or via e-mail after all participants completed the POST assessment to arrange a time and date to complete a satisfaction survey.
PCPs
The provider version of the SCPs was sent to the PCPs of the participants who had given permission to share their information. If a PCP was within the hospital network, he or she was e-mailed the SCP and satisfaction survey. If a PCP was outside the network, he or she was sent the SCP via secure fax and contacted via phone by the principal investigator to complete the satisfaction survey.
Results
Data Analytic Plan
Descriptive statistics were determined with demographic data. Means and standard deviations (SDs) were calculated for satisfaction ratings and PLANS scores – pre and post. Wilcoxon signed rank tests were also used for tests of significance. Finally, percentages were calculated for OCP, PCP, and patient follow-up data related to evaluation of the POST program.
Participant Demographics
Twenty-five patients with breast cancer were enrolled in the field test. The mean age at the time of enrollment was 61 years (SD, 13.59), 92% were Caucasian, and 8% were black or African American. No participants were Hispanic or Latina. Three women were enrolled prior to ending active treatment but were within a close enough proximity that the OCP felt they were ready for an SCP. Thirteen women were enrolled within 6 months, and 9 were enrolled within 1 year after ending active treatment for breast cancer.
Patient Outcomes
Baseline and Postintervention Confidence
Ratings – PLANS
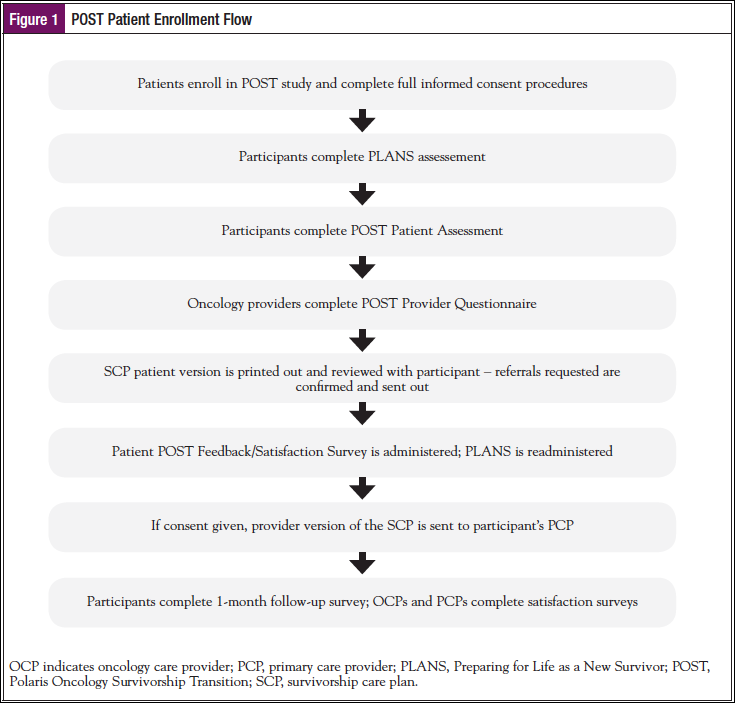
Twenty-three patients (92.0%) completed the PLANS before taking the patient assessment and after reviewing their SCP. Confidence ratings were equally high before and after the patient assessment (Figure 2). At both times, the median of the 5-item average score was 9.40 (range, 6.60-10.00) on a 10-point scale where 10 = “Extremely Confident,” Z = 0.31, P = .75, and r = .06. Immediately following the POST, 84% of patients had an average score of 8 or higher. At follow-up, 91% had similar scores. Ratings of surveillance and coordination were generally high before the Patient Assessment, with a median score of 3.00 on the 4-point scale, where higher scores indicated more favorable responses (range, 2.45-4.00). However, a Wilcoxon signed rank test indicated that scores were significantly higher after assessment and review of the SCP (median, 3.45; range, 2.36-4.00), Z = 2.03, P = .04, and r = .42.
Baseline Feedback/Satisfaction of Patient
Assessment
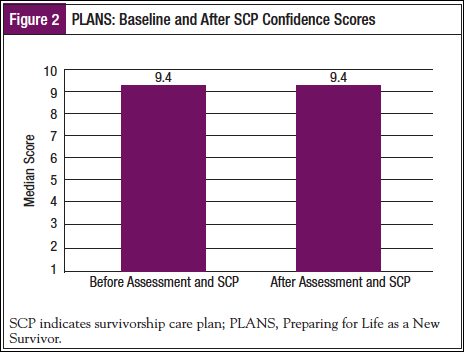
Twenty-five patients rated the Patient Assessment immediately after completing it. Sixty-eight percent of the patients were “Very Satisfied” with the assessment process overall, 28.0% were “A Lot Satisfied,” and 4.0% were “A Little Satisfied.” Figure 3 provides specific details regarding respondent ratings.
Patient Baseline Feedback/Satisfaction of SCP
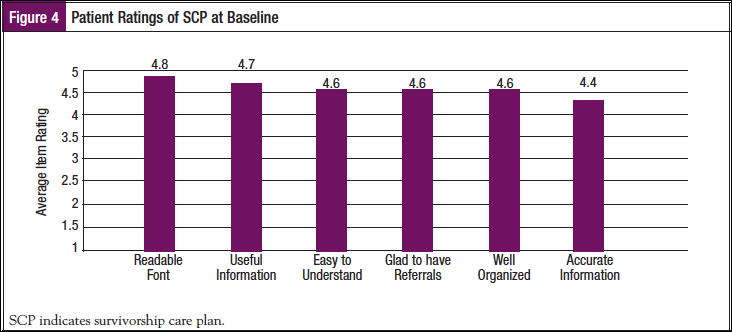
Twenty-three patients rated the SCP. Seventy percent (69.6%) of the patients were “Very Satisfied” with the SCP overall, 26.1% were “A Lot Satisfied,” and 4.3% were “A Little Satisfied.” Figure 4 provides specific details regarding respondent ratings.
Patient Follow-up
One-month follow-up assessments were completed with 22 of 25 participants (88.0%) over the phone. Two participants were lost to follow-up and 1 withdrew from the study. Questions were posed regarding usefulness of the SCP as well as relevant behavioral changes since baseline. Wilcoxon signed rank tests indicated that satisfaction scores were not significantly different at follow-up compared with ratings immediately following the SCP review (patient assessment, Z = –0.71, P = .48, r = –0.15; SCP, Z = –0.82, P = .41, r = –0.17). Table 2 provides additional information about patient feedback at follow-up.
OCP Outcomes
Three oncologists and 1 oncology nurse practitioner completed the satisfaction survey asking about the Provider Questionnaire and SCPs. None had prior formal experience using an SCP in their clinical practice, but all were familiar with existing options such as Journey Forward. See Table 3 for information about OCP feedback.
PCP Outcomes
Although all PCPs confirmed receipt of the SCP, only 9 of 21 PCPs (42.8%) completed the online satisfaction survey. Therefore, we present their data here to be interpreted with caution. Overall, 44.4% of PCPs were “Very Satisfied” with the POST program, 33.3% were “A Lot Satisfied,” and 22.2% were “Somewhat” or “A Little Satisfied.” See Table 3 for information about PCP feedback.
Discussion
The POST is innovative and may be a good resource for providers who are striving to meet the standards surrounding survivorship care planning. First, the POST produces tailored survivorship plans that fully reflect IOM recommendations and ACS requirements. Second, the content and crafting of the assessment and SCP were heavily informed by patients, OCPs, and PCPs. Third, the POST program has the capacity to communicate with the EHR, which significantly impacts efficiency in building survivorship plans. However, it should be noted that the EHR integration was developed after the end of this study and is now part of the POST program during our ongoing, phase 2, NCI-funded, randomized controlled trial. Fourth, the POST is both patient and provider driven, whereas most existing programs are simply provider driven. Fifth, the SCPs include a thorough medical summary and plan as well as a psychosocial summary of functioning, and suggestions to improve survivorship functioning over time. Finally, the POST aids communication and the transfer of care from the oncology team back to the PCP by sending a copy of the SCP to the patients’ PCPs. Given that many PCPs do not receive detailed information, and in the worst situation do not receive any information from the oncology team, the POST’s automatic transmission of the SCP to the PCP is pioneering.
A key point of this study was clinical implementation, as all SCPs were built in “real time” during the clinic visits. We wanted the study to mirror real-life clinical practice; however, because this was a development and field trial project, we had to rely primarily on the research staff as we were working through programming development iterations while conducting the field trial. The oncology care team was involved with building the medical sections of the SCP and was the group who reviewed the care plans for accuracy. In our ongoing phase 2 study, we have built a protocol that mirrors what we believe is a viable option for oncology treatment centers to do wide-scaled survivorship planning.
In our protocol, the nurse practitioners/oncology staff build the care plans, including information prepopulated from the EHR, prior to the patient coming in for a “survivorship care planning” visit. There, the plan is reviewed with the patient by a nurse practitioner. After the review, the patient sees her oncologist for 1 final visit during which she has an opportunity to ask questions and address any concerns or information provided in the SCP. The provider then signs off on the SCP, and the briefer provider version of the SCP is both uploaded to the EHR and sent to the PCP. Finally, the patient retains a physical copy of the patient version of the SCP. Results from this study will be disseminated once the study is complete in 2018.
Another important piece of SCP implementation is how to handle patients who have psychosocial needs that are identified in their assessments. For this present study, we were able to make referrals to the on-site psychologist and social worker, who would then contact patients to schedule appointments or visit the clinic if it was an urgent situation. This worked well, and we found that it was very helpful for the psychologist and social worker to be able to review the SCP prior to seeing the patient. However, there are some situations where oncology clinics do not have psychologists on staff to address psychological needs of patients. In these situations, we feel technology can, again, be the answer to connecting patients with resources in their communities. For example, in a different oncology care–related project, our team built the technology to identify psychosocial distress in patients with cancer through a patient-driven, web-based program that could also match a patient’s insurance and zip code to a tailored database we built.15 This aided patients who were not interested in seeing a provider in the cancer center for various reasons. Polaris Health Directions, the technology team that built the POST, has the ability to integrate a similar type of referral system into the POST program.
We assessed both OCP and PCP outcomes, as we were interested in their perceptions of the clinical utility of the POST program. Although we do not present the data here since the number of providers sampled is very low (OCP = 4, PCP = 9), there was a trend that the OCP felt the SCP was most helpful in building and presenting the medical diagnosis and treatment summary as well as the medical care plan. Further, the majority of both the OCP and PCP endorsed that they would consider using the SCP in clinical practice and would recommend the SCP to colleagues for survivorship planning. Finally, the PCPs were most likely to report that they felt that having the SCP would benefit clinical interactions with patients, and that the SCP helped stimulate conversation that would not otherwise have come up in their usual interactions with patients. However, the PCPs did not feel that the SCP impacted their decision-making in their clinical encounters.
We also assessed patient ratings regarding confidence for survivorship, before and after review of the SCP. As shown in Figure 2, patient ratings of confidence did not change over time. This was likely because the sample’s confidence pre-SCP was very high, which allows for little improvement post-SCP. Figures 3 and 4 provide information about how the patients viewed the assessment and the actual SCP that was generated from the POST program, and it appears the patients had quite favorable reviews of both. It should also be noted that patient ratings of the program remained favorable at the 1-month follow-up (Table 2).
There are many limitations to this study. The most important limitation is that this study does not show that the POST program is any more efficient than the other planning programs in existence. We were unable to achieve the EHR integration when we were conducting the field study and were busy working on the development and testing of the POST program. However, it should be noted that the purpose of a phase 1 STTR is to develop and test programs like the POST, so that by the end of the study there is a streamlined, usable program to test in a phase 2 trial where issues like efficiency can be addressed. We presently have the phase 2 randomized controlled trial under way and are measuring time to build the SCP compared with other programs, as we feel that the true innovation of the POST 2 is its efficacy and ability to integrate into real clinical practices.
There is much we still do not know about survivorship care planning—its impact on patients, and its impact on clinical practices. Research is needed to determine the potential impact of survivorship planning on health and psychosocial outcomes. Mayer et al8 suggested 4 broad areas of SCP research: content, dissemination and implementation, outcomes, and improved study methodology. We need a better understanding of issues that are important to cancer survivors, as well as what strategies we can use to best prepare patients for survivorship.16
In terms of dissemination and implementation, there is a great need for studies aimed at developing guidelines for institutional use so cancer centers can comply with IOM recommendations.6 We need a better understanding of best possible reimbursement pathways, and optimal insurance and payment options.6 Identifying the best way for OCPs to receive compensation for SCP delivery is essential.17 Also, as addressed above, a consistent barrier to SCP implementation is lack of time. Developing and delivering an SCP could take between 1 and 4 hours per patient.18 This time demand can be reduced by integrating planning programs with the EHR or cancer registry. This will be an important piece of future studies as we continue to investigate the best strategies, and potential consequences and benefits, of EHR or cancer registry integration.19
Outcomes research related to SCPs should focus on how to best measure important constructs or difficulties experienced by patients.14 We did not present patient outcomes in this paper as our sample was small and the purpose of the field test was POST program development and utility. Because there is a lack of randomized controlled trials in this area of study, it is difficult to confidently say which issues remain important to the quality of life of survivors over time, and sound methodological studies are needed to determine the important constructs that threaten the well-being of survivors. Finally, more investigation is needed regarding how SCPs can best facilitate communication between medical providers.17 Gaps in physician understanding must be thoroughly defined to help guide SCP content, and PCP training in survivorship planning may be important for improving care as it transfers from the oncology team back to general medical practice.20
References
- DeSantis CE, Lin CC, Mariotto AB, et al. Cancer treatment and survivorship statistics, 2014. CA Cancer J Clin. 2014;64:252-271.
- American College of Surgeons. Commission on Cancer. Cancer Program Standards 2012: Ensuring Patient-Centered Care. V1.2.1. www.facs.org/~/media/files/quality%20programs/cancer/coc/programstandards2012.ashx. Accessed September 1, 2015.
- Grunfeld E, Earle CC. The interface between primary care and oncology specialty care: treatment through survivorship. J Natl Cancer Inst Mongr. 2010;2010:25-30.
- Hewitt M, Greenfield S, Stoval E. From Cancer Patient to Cancer Survivor: Lost in Transition. Washington, DC: The National Academies Press; 2006.
- Smith SL, Wai ES, Alexander C, et al. Caring for survivors of breast cancer: perspective of the primary care physician. Curr Oncol. 2011;18:e218-e226.
- Stricker CT, Jacobs LA, Risendal B, et al. Survivorship care planning after the Institute of Medicine recommendations: how are we faring? J Cancer Surviv. 2011;5:358-370.
- Stricker CT, O’Brien M. Implementing the Commission on Cancer standards for survivorship care plans. Clin J Oncol Nurs. 2014;18(suppl):15-22.
- Mayer DK, Birken SA, Check DK, et al. Summing it up: an integrative review of studies of cancer survivorship care plans (2006-2013). Cancer. 2015;121:978-996.
- Grunfeld E, Julian JA, Pond G, et al. Evaluating survivorship care plans: results of a randomized, clinical trial of patients with breast cancer. J Clin Oncol. 2011;29:4755-4762.
- Jefford M, Schofield P, Emery J. Improving survivorship care. J Clin Oncol. 2012;30:1391-1392.
- Smith TJ, Snyder C. Is it time for (survivorship care) plan B? J Clin Oncol. 2011;29:4740-4742.
- Dunn RL, Crowley SA, Janz NK, et al. Impact of a transition visit on addressing quality of life and readiness to assume greater self-management among breast cancer survivors. Psychooncology. 2011;20:S91.
- National Cancer Institute. National Institutes of Health. GEM Grid-Enabled Measure Database. Preparing for Life as a New Survivor (PLANS). gem-beta.org/public/MeasureDetail.aspx?cat=2&mid=1391&tab=0. Accessed October 6, 2016.
- Brothers BM, Easley A, Salani R, et al. Do survivorship care plans impact patients’ evaluations of care? A randomized evaluation with gynecologic oncology patients. Gynecol Oncol. 2013;129:554-558.
- O’Hea EL, Cutillo A, Dietzen L, et al. Randomized control trial to test a computerized psychosocial cancer assessment and referral program: methods and research design. Contemp Clin Trials. 2013;35:15-24.
- Parry C, Kent EE, Forsythe LP, et al. Can’t see the forest for the care plan: a call to revisit the context of care planning. J Clin Oncol. 2013;31:2651-2653.
- Burg MA, Lopez ED, Dailey A, et al. The potential of survivorship care plans in primary care follow-up of minority breast cancer patients. J Gen Intern Med. 2009;24(suppl 2):S467-S471.
- Brennan ME, Gormally JF, Butow P, et al. Survivorship cancer plans in cancer: a systematic review of care plan outcomes. Br J Cancer. 2014;111:1899-1908.
- Dulko D, Pace CM, Dittus KL, et al. Barriers and facilitators to implementing cancer survivorship plans. Oncol Nurs Forum. 2013;40:575-580.
- Forsythe LP, Parry C, Alfano CM, et al. Use of survivorship care plans in the United States: associations with survivorship care. J Natl Cancer Inst. 2013;105:1579-1587.




