Background: Despite the evidence establishing screening as effective in decreasing cancer mortality, rates remain below recommended levels. Patient navigation (PN) has shown promise to increase screening, yet the strength of the evidence of PN on screening rates is unknown.
Objectives: To determine the effects of PN, including professional navigators (nurse and non-nurse) versus trained lay navigators, on all cancer screening behaviors.
Methods: Medical and social science databases were searched for randomized controlled trials published in English between 1989 and May 2015 for inclusion in a meta-analysis. The review process was guided by PRISMA. Data were extracted to assess the effect of navigation on cancer screening rates. Random effects models were used to compute risk ratios, and I2 statistics determined the impact of heterogeneity.
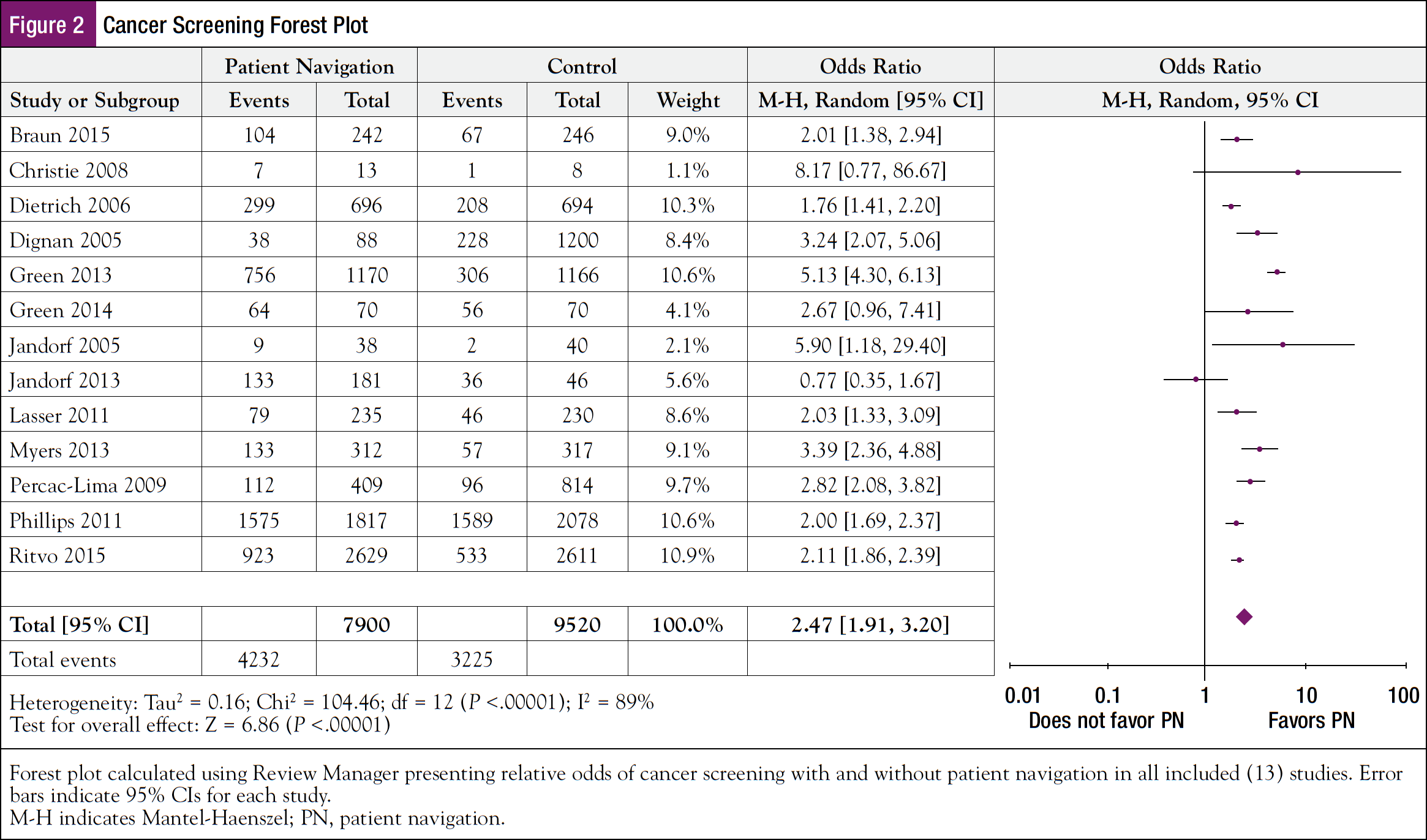
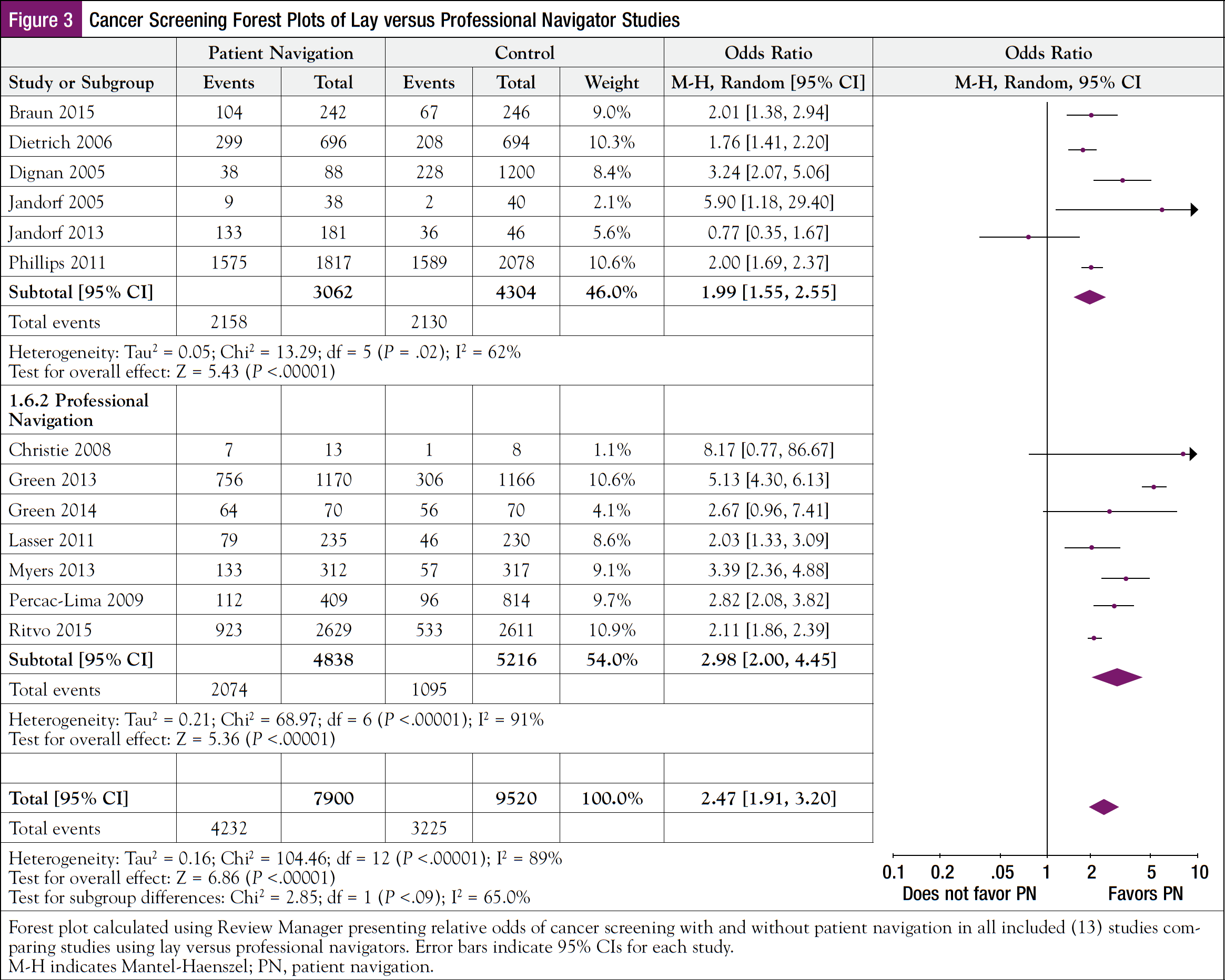
Results: Of 3985 articles screened, 13 articles examining the effect of PN on cancer screening were included in this analysis. Compared with usual care, patients who received PN were significantly more likely to access cancer screening (odds ratio [OR], 2.47; 95% CI, 1.91-3.20; P <.00001), and both professional (OR, 2.98; 95% CI, 2.00-4.45; P <.0001) and lay (OR, 1.99; 95% CI, 1.55-2.55; P <.0001) navigators had a significant effect on cancer screening rates.
Discussion: The results suggest that PN is an effective intervention for increasing cancer screening, with greater effects achieved by professional navigators.
Conclusion: The evidence provided by this meta-analysis supports the implementation of nurse navigation programs to promote screening rates.
Cancer screening can significantly reduce mortality rates related to breast cancer,1,2 colorectal cancer,3 and cervical cancer4. Various cancer screening tools have been proved effective in detecting cancers, including mammography for breast cancer,5 pap tests for cervical cancer,6 and fecal occult blood testing, sigmoidoscopy, colonoscopy, and fecal immunochemical test for colorectal cancer,7,8 all of which are recommended by both the American9 and Canadian10 cancer societies. Due in large part to increases in cancer screening rates over the past 2 decades, rates of cancer mortality have been falling.11,12 However, despite this promising trend, cancer screening rates remain below the recommended levels,13 especially among ethnic minorities,13,14 those with low socioeconomic status,15-18 and the uninsured.17,19,20 Additionally, many patients experience a variety of barriers to cancer screening, including lack of awareness of the need for screening,18,21 lack of recommendation from a physician,21 language barriers, logistical challenges to attending screening sessions, cultural beliefs regarding cancer, negative views of cancer and cancer screening, and lack of motivation for screening.18
Patient navigation (PN) was introduced in the 1990s in Harlem, NY, as an intervention to address barriers to cancer care access.22,23 PN is defined as a one-on-one interaction between a navigator and a patient. The navigator, either a professional, such as a nurse, or a trained layperson, provides health education and support to a patient to promote recommended healthcare utilization behaviors from screening to diagnosis to resolution.24,25 Traditionally, PN intervention programs have trained and employed lay individuals as navigators who are representative of an affected community, who have experience with a particular disease/condition (eg, cancer), and who share a community of identity (eg, geography, race/ethnicity, culture, language, gender). Navigators have traditionally been non–health professionals, or peer helpers, using firsthand knowledge and experience with the healthcare system to benefit others.23 However, in the past 25 years since PN interventions were introduced into research and practice settings, the navigator role has expanded to include trained healthcare professionals, such as nurses,26 otherwise referred to as professional navigators.
The current paper presents the results of an analysis of the evidence for PN effectiveness to improve cancer screening rates. These results are part of a larger meta-analysis conducted to examine the effectiveness of PN as an intervention on healthcare utilization behavior outcomes in both cancer and noncancer patient populations, the results of which have been previously published.27 Our previous meta-analysis included an analysis of PN effectiveness for increasing general health screening rates. Here, we present our findings regarding the effectiveness of PN, provided by professional and lay navigators, specifically for improving cancer screening rates.
Methods
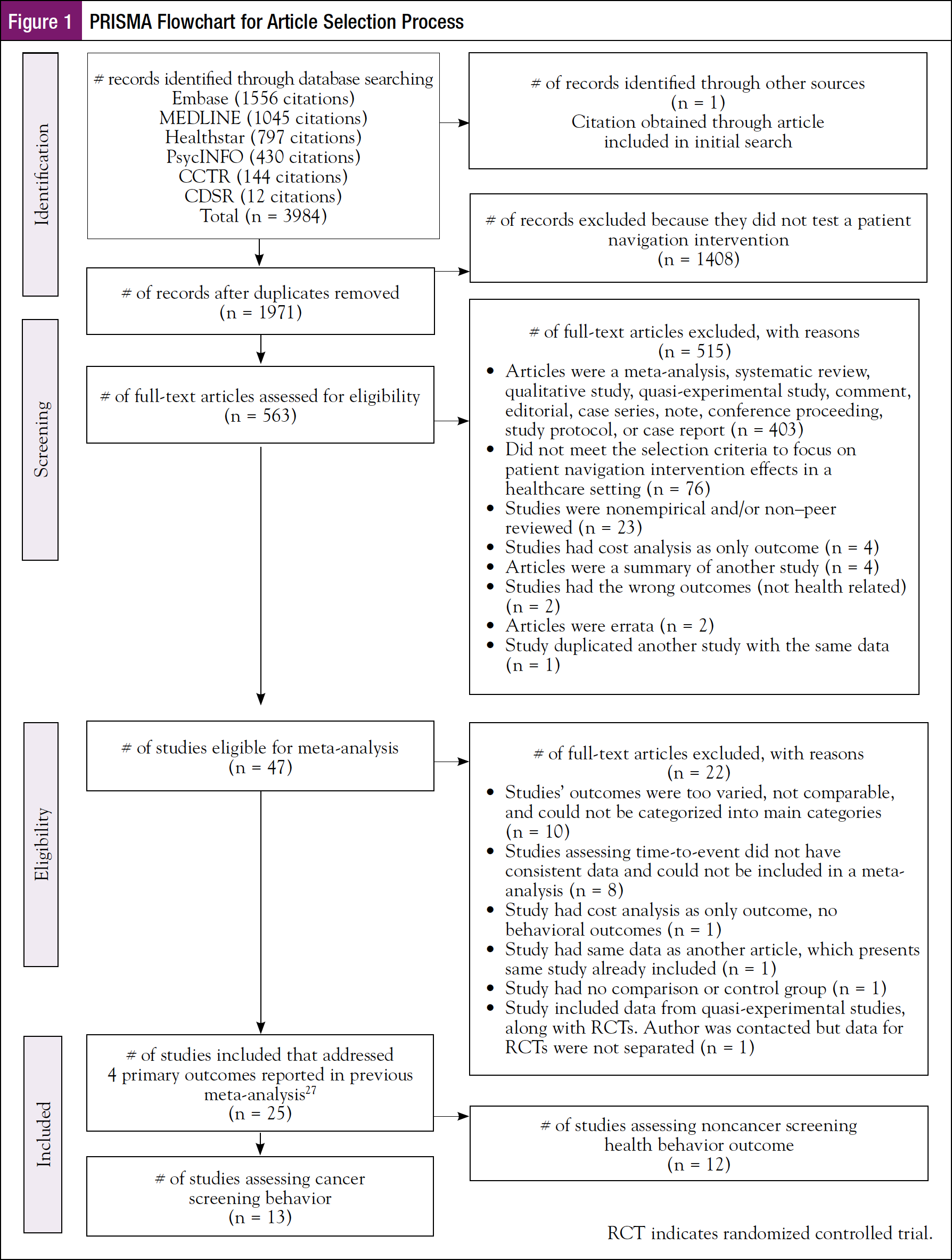
Following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement and recommendations28 as a guide for the development and completion of this meta-analysis, we began with comprehensive literature searches of medical and social science databases including MEDLINE, PsycINFO, EBM Reviews – Cochrane Central Register of Controlled Trials, Healthstar, Joanna Briggs Institute EBP database, and Embase for research published between 1989 and May 2015. Citations from all databases (N = 3985) were reviewed, and duplicates (n = 2014) were removed in Covidence.org. Two authors (SAF, TC) independently reviewed the remaining abstracts (n = 1971) for eligibility. Discrepancies regarding the inclusion or exclusion of studies were resolved by a third author (LBS). Included studies had to be randomized controlled trials (RCTs) with a comparison group; use the term “navigation” in the description of the intervention (eg, navigator, navigate); test a PN intervention, measure the intervention components, and assess a health outcome; be published in a peer-reviewed journal; and be available in the English language. Papers were excluded if studies were quasi-experimental, qualitative, case studies, nonempirical, non–peer-reviewed, or testing technology applications; were meta-analyses, systematic reviews, comments, editorials, case series, notes, or conference proceedings. For the current analysis, only those studies assessing completion of a cancer screening behavior were included. Figure 1 presents the PRISMA flowchart outlining the process of article selection.
Two authors (SAF, NMJ) independently extracted data, including nature of navigation (lay vs professional) and results from the RCT, which were entered into an Excel database and subsequently verified for any discrepancies. Data analysis was completed using Review Manager 5.3.29 The random effects model was used to examine combined results due to the anticipated clinical heterogeneity in the methodology of studies. Dichotomous outcome measures for each RCT
were expressed as odds ratios (ORs) and 95% confidence intervals (CIs). To determine the impact of heterogeneity on the meta-analysis, I2 statistics were used. An I2 score of ≤40% was considered unimportant heterogeneity.30 For each study included in a model, the effect was plotted according to the inverse of its standard error. Egger’s regression intercept of funnel plot asymmetry was computed to test the presence of publication bias.31
Results
Of the 3985 articles with the potential of being eligible for inclusion, 47 full text articles were considered for eligibility. Further review resulted in the removal of 22 articles owing to factors such as not assessing a health behavior outcome (ie, cost analysis); assessing outcomes that could not be categorized for a meta-analysis; lacking a comparison or control group, including a secondary analysis of subjects and intervention, and including data combined from RCT and quasi-experimental methods. The final pool consisted of 25 studies, which are reported elsewhere,27 and focused on both cancer and noncancer patient populations and health outcomes. The 13 studies selected for this analysis exclusively focused on cancer screening behaviors, including mammograms, pap tests, colonoscopies, fecal occult blood tests, and endoscopies.32-44
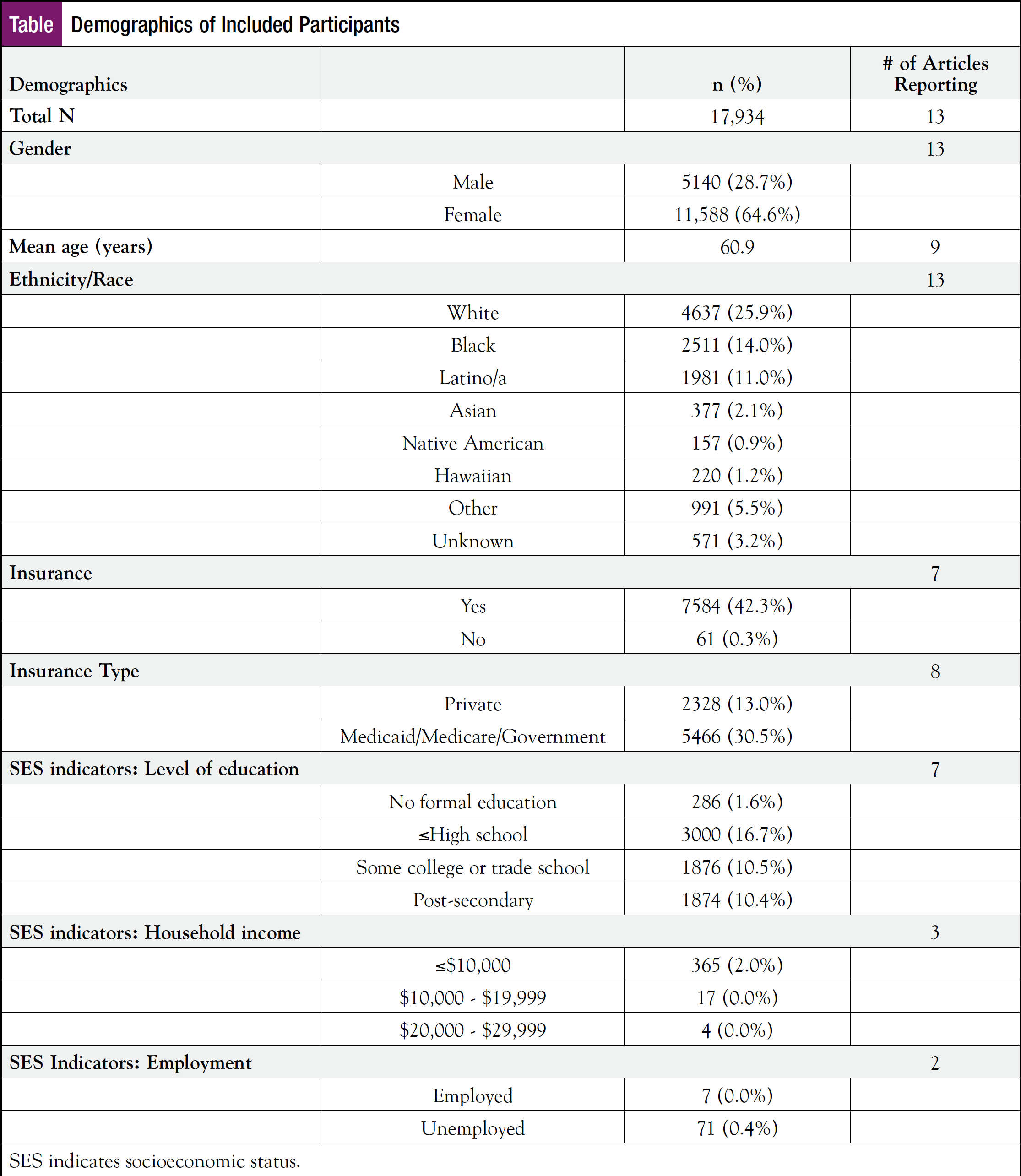
The selected studies included data collected between years 2005 and 2015. The 13 included studies had a sample size of 17,934 participants, with 8412 allocated to a PN intervention and 9522 to the usual care condition. The majority of participants were women (n = 11,588; 64.6%), whereas less than one-third of participants were male (n = 5140; 28.7%). White patients comprised 25.9% of the participant population, followed by black and Latino/a participants at 14.0% and 11.0%, respectively. Mean ages of participants ranged from 54.4 to 67.6 years, with an overall mean age of 60.9 years. Six studies (46.2%) included multisite testing, and the majority (92.3%) were conducted in the United States, with 1 in Canada (7.7%). The Table presents the demographics of included participants. Follow-up time to assess the effectiveness of PN ranged from 2 to 3 weeks to 18 months, with a mean time of 28.9 weeks, including second and third follow-up points. One study (7.7%) tested PN in an academic hospital setting, and 38.5% assessed PN in a community health setting. However, it was unclear for more than 50% of the studies whether they were in an academic or community setting as it was not explicitly stated. Finally, slightly less than half (46.2%) of the studies reported details on how navigators were trained32,35,38,40,42,43 to varying degrees of detail.
Pooled analysis revealed that, significantly, patients who received PN were almost 2.5 times as likely to access health screening (OR, 2.47; 95% CI, 1.91-3.20; P <.00001) (Figure 2) than those who did not receive PN. PN was delivered by trained laypersons in 5 studies (38.5%),32,34,35,38,43 professional navigators in 7 (53.8%) with 5 professional navigators being nurses (38.5%),33,36,37,40,42,44 and a combination of lay and professional navigators in 1 (7.7%) study.35 A subgroup analysis comparing trained lay navigators with professional navigators revealed significant evidence that the effect size was related to type of support provided (P = .00001). As our analysis included very few non-nurse professional navigators (eg, outreach workers), both nurse and non-nurse navigators were identified as professional navigators. Both professional (OR, 2.98; 95% CI, 2.00-4.45; P <.0001) and lay (OR, 1.99; 95% CI, 1.55-2.55; P <.0001) navigators had a significant effect on cancer screening rates (Figure 3).
Discussion
The results from this meta-analysis suggest that PN is an effective intervention for increasing cancer screening behaviors. The pooled analysis revealed that PN increased the likelihood of cancer screening uptake by almost 2.5 times compared with usual care, making it an effective intervention for a variety of cancer screenings, including mammograms, pap tests, colonoscopies, fecal occult blood tests, and endoscopies. These results also provide support for the use of both trained lay and professional navigators, such as nurses, to increase cancer screening rates, although professional navigators appeared to show more promise.
The majority of participants analyzed in the studies were women (64.6%), indicating that PN is an effective intervention for women. However, the relatively small number of men (28.7%) in these studies means that the effect of PN on screening rates among men is largely unknown, highlighting the need to further test PN with male cancer patient populations. With regard to ethnicity, one-quarter of participants in the studies were white (25.9%), while the remaining were black (14.0%), Latino/a (11.0%), Asian (2.1%), Hawaiian (1.2%), Native American (0.9%), Other (5.5%), and Unknown (3.2%), suggesting PN to be an effective intervention for increasing cancer screening among ethnic minority populations, although further investigation of this relationship is required.
The limitations of this meta-analysis warrant caution when interpreting these results. First, the degree of heterogeneity was high in this meta-analysis with an I2 value of 89%, indicating great diversity in effect sizes, thus complicating the interpretation of the results. Second, follow-up times (the point at which outcomes were assessed) varied greatly between studies with times ranging anywhere from 2 to 18 months. Third, PN interventions were tested in diverse settings (eg, community health centers, hospitals) and lacked standardization, and the modalities of PN intervention were also diverse in content and delivery. The nature of interactions between navigators and patients necessitates a certain level of flexibility due to the unique needs of each patient, yet flexibility increases the difficulty of controlling for confounding factors. However, the standardization of PN interventions and training is necessary to reduce the possibility of confounding factors. Finally, details of intervention content and navigator training were unclear, thereby making it difficult to draw conclusions regarding which components of PN were effective for increasing cancer screening.
Conclusion
The current literature on the effectiveness of PN for cancer screening behaviors demonstrates that PN is a successful intervention for increasing cancer screenings, with both trained lay and professional navigators, including nurses, demonstrating effectiveness. Both women and ethnic minorities benefit from PN, whereas the effects of PN for men need further examination. Yet, given that the interventions tested varied considerably, identifying successful elements of PN interventions becomes difficult. However, as research has already established a relationship between increased cancer screening and lower rates of mortality,1-4 the implementation of PN as a health behavior change intervention in the healthcare system may have positive implications for mortality rates in cancer populations. Although further research is required to understand how the various components of PN are effective, the results from the current meta-analysis support PN as an effective intervention for increasing cancer screening rates.
Acknowledgments
We would like to thank Maureen Pakosh, Information Specialist with Library & Information Services, University Health Network Toronto Rehab in Toronto, Canada, for conducting our search of the databases.
Compliance with Ethical Standards
Conflict of Interest: The authors declare that they have no conflict of interest.
Ethical approval: This article does not contain any studies with human participants performed by any of the authors.
References
- Smith RA, Duffy SW, Gabe R, et al. The randomized trials of breast cancer screening: what have we learned? Radiol Clin North Am. 2004;42:793-806.
- Humphrey LL, Helfand M, Chan BK, et al. Breast cancer screening: a summary of the evidence for the U.S. Preventive Services Task Force. Ann Intern Med. 2002;137(5 Pt 1):347-367.
- Hewitson P, Glasziou P, Irwig L, et al. Screening for colorectal cancer using the faecal occult blood test, Hemoccult. Cochrane Database Syst Rev. 2007;(1):CD001216.
- Vesco KK, Whitlock EP, Eder M, et al. Screening for Cervical Cancer: A Systematic Evidence Review for the U.S. Preventive Services Task Force. Evidence Syntheses No. 86. AHRQ Publication No. 11-05156-EF-1. Rockville, MD; 2011.
- Nelson HD, Cantor A, Humphrey L, et al. Screening for Breast Cancer: A Systematic Review to Update the 2009 U.S. Preventive Services Task Force Recommendation. Evidence Syntheses No. 124. AHRQ Publication No. 14-05201-EF-1. Rockville, MD; 2016.
- Whitlock EP, Vesco KK, Eder M, et al. Liquid-based cytology and human papillomavirus testing to screen for cervical cancer: a systematic review for the U.S. Preventive Services Task Force. Ann Intern Med. 2011;155:687-697.
- Levin B, Lieberman DA, McFarland B, et al. Screening and surveillance for the early detection of colorectal cancer and adenomatous polyps, 2008: a joint guideline from the American Cancer Society, the US Multi-Society Task Force on Colorectal Cancer, and the American College of Radiology. Gastroenterology. 2008;134:1570-1595.
- Whitlock EP, Lin JS, Liles E, et al. Screening for colorectal cancer: a targeted, updated systematic review for the U.S. Preventive Services Task Force. Ann Intern Med. 2008;149:638-658.
- American Cancer Society. American Cancer Society Guidelines for the Early Detection of Cancer. 2016. www.cancer.org/healthy/find-cancer-early/cancer-screening-guidelines/american-cancer-society-guidelines-for-the-early-detection-of-cancer.html. Accessed October 14, 2016.
- Canadian Cancer Society. Screening. 2016. www.cancer.ca/en/prevention-and-screening/early-detection-and-screening/screening/?region=on. Accessed October 16, 2016.
- Byers T. Two decades of declining cancer mortality: progress with disparity. Annu Rev Public Health. 2010;31:121-132.
- Kohler BA, Sherman RL, Howlader N, et al. Annual report to the nation on the status of cancer, 1975-2011, featuring incidence of breast cancer subtypes by race/ethnicity, poverty, and state. J Natl Cancer Inst. 2015;107:djv048.
- Centers for Disease Control and Prevention. Cancer screening – United States, 2010. MMWR Morb Mortal Wkly Rep. 2012;61:41-45.
- Monnat SM. Race/ethnicity and the socioeconomic status gradient in women’s cancer screening utilization: a case of diminishing returns? J Health Care Poor Underserved. 2014;25(1):332-356.
- Breen N, Wagener DK, Brown ML, et al. Progress in cancer screening over a decade: results of cancer screening from the 1987, 1992, and 1998 National Health Interview Surveys. J Natl Cancer Inst. 2001;93:1704-1713.
- Link BG, Northridge ME, Phelan JC, et al. Social epidemiology and the fundamental cause concept: on the structuring of effective cancer screens by socioeconomic status. Milbank Q. 1998;76:375-402.
- Rosenberg L, Wise LA, Palmer JR, et al. A multilevel study of socioeconomic predictors of regular mammography use among African-American women. Cancer Epidemiol Biomarkers Prev. 2005;14(11 Pt 1):2628-2633.
- Honein-AbouHaidar GN, Kastner M, Vuong V, et al. Systematic review and meta-study synthesis of qualitative studies evaluating facilitators and barriers to participation in colorectal cancer screening. Cancer Epidemiol Biomarkers Prev. 2016;25:907-917.
- Meyer CP, Allard CB, Sammon JD, et al. The impact of Medicare eligibility on cancer screening behaviors. Prev Med. 2016;85:47-52.
- Robinson JM, Shavers V. The role of health insurance coverage in cancer screening utilization. J Health Care Poor Underserved. 2008;19:842-856.
- Finney Rutten LJ, Nelson DE, Meissner HI. Examination of population-wide trends in barriers to cancer screening from a diffusion of innovation perspective (1987-2000). Prev Med. 2004;38:258-268.
- Freeman HP. Patient navigation: a community centered approach to reducing cancer mortality. J Cancer Educ. 2006;21(suppl):S11-S14.
- Freeman HP. Patient navigation: a community based strategy to reduce cancer disparities. J Urban Heal. 2006;83:139-141.
- Freeman HP, Rodriguez RL. History and principles of patient navigation. Cancer. 2011;117(suppl S15):3539-3542.
- Lorhan S, Cleghorn L, Fitch M, et al. Moving the agenda forward for cancer patient navigation: understanding volunteer and peer navigation approaches. J Cancer Educ. 2013;28:84-91.
- Freeman HP. The history, principles, and future of patient navigation: commentary. Semin Oncol Nurs. 2013;29:72-75.
- Ali-Faisal SF, Colella TJ, Medina-Jaudes N, et al. The effectiveness of patient navigation to improve healthcare utilization outcomes: a meta-analysis of randomized controlled trials. Patient Educ Couns. 2017;100:436-448.
- Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA Statement. PLoS Med. 2009;6:e1000097.
- Review Manager (RevMan). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cohrane Collaboration, 2014.
- Deeks JJ, Higgins JP, Altman DG. Analysing data and undertaking meta-analyses. In: Higgins JP, Green S, eds. Cochrane Handbook for Systematic Reviews of Interventions. The Cochrane Series. Chichester, West Sussex: John Wiley & Sons, Ltd; 2008:278.
- Egger M, Davey Smith G, Schneider M, et al. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629-634.
- Braun KL, Thomas WL Jr, Domingo JL, et al. Reducing cancer screening disparities in medicare beneficiaries through cancer patient navigation. J Am Geriatr Soc. 2015;63:365-370.
- Christie J, Itzkowitz S, Lihau-Nkanza I, et al A randomized controlled trial using patient navigation to increase colonoscopy screening among low-income minorities. J Natl Med Assoc. 2008;100:278-284.
- Dietrich AJ, Tobin JN, Cassells A, et al. Telephone care management to improve cancer screening among low-income women. Ann Intern Med. 2006;144:563-571.
- Dignan MB, Burhansstipanov L, Hariton J, et al. A comparison of two Native American navigator formats: face-to-face and telephone. Cancer Control. 2005;12(suppl 2):28-33.
- Green B, Wang CY, Anderson ML, et al. An automated intervention with stepped increases in support to increase uptake of colorectal cancer screening. Ann Intern Med. 2013;158(5 Pt 1):301-311.
- Green BB, Anderson ML, Wang CY, et al. Results of nurse navigator follow-up after positive colorectal cancer screening test: a randomized trial. J Am Board Fam Med. 2014;27:789-795.
- Jandorf L, Gutierrez Y, Lopez J, et al. Use of a patient navigator to increase colorectal cancer screening in an urban neighborhood health clinic. J Urban Health. 2005;82:216-224.
- Jandorf L, Braschi C, Ernstoff E, et al. Culturally targeted patient navigation for increasing African Americans’ adherence to screening colonoscopy: a randomized clinical trial. Cancer Epidemiol Biomarkers Prev. 2013;22:1577-1587.
- Lasser KE, Murillo J, Lisboa S, et al. Colorectal cancer screening among ethnically diverse, low-income patients: a randomized controlled trial. Arch Intern Med. 2011;171:906-912.
- Myers RE, Bittner-Fagan H, Daskalakis C, et al. A randomized controlled trial of a tailored navigation and a standard intervention in colorectal cancer screening. Cancer Epidemiol Biomarkers Prev. 2013;22:109-117.
- Percac-Lima S, Grant RW, Green AR, et al. A culturally tailored navigator program for colorectal cancer screening in a community health center: a randomized, controlled trial. J Gen Intern Med. 2009;24:211-217.
- Phillips CE, Rothstein JD, Beaver K, et al. Patient navigation to increase mammography screening among inner city women. J Gen Intern Med. 2011;26:123-129.
- Ritvo PG, Myers RE, Paszat LF, et al. Personal navigation increases colorectal cancer screening uptake. Cancer Epidemiol Biomarkers Prev. 2015;24:506-511.




