Background: Of the 1.6 million individuals annually diagnosed with cancer, 522,166 received chemoradation, with 234,975 developing toxic mucositis and 20% dying within 5 years due largely to mucositis-mediated unplanned treatment breaks. In a mucositis registry study1 of 66 consecutive patients undergoing chemoradiation using 2 different radiation protocols, 5 targeted immuno and 6 nontargeted chemotherapies by 32 oncologists in 24 institutions across 14 states in the United States, ProThelial (polymerized cross-linked sucralfate) was prescribed for prevention in 8 patients and for elimination in 58 patients (48 males, 18 females, aged 14-96 years). All 8 patients treated for prevention, 53 patients treated for reversal, and 5 patients lost to follow-up entered into data analysis. Primary outcomes of patient-reported mouth/throat soreness, and clinician-observed WHO grade mucositis, EROTC/RTOG-oral and GI mucositis were assessed at days 1, 2, 3, and 7 of use of ProThelial and at the close of cancer treatment. All 8 patients scheduled for G-tube and expected to develop mucositis never developed mucositis and did not require G-tube placement. All 53 patients had 2- to 3-day elimination of mucositis sustained throughout treatment as assessed by both primary outcome measures.
Objectives: Grade the significance of the ProThelial data through a statistical analysis of treatment effect compared with historical controls of 351 patients experiencing mucositis duration of 70 to 84 days.2-4
Methods: Complete a Glasziou rate ratio analysis,5 a relative risk analysis, and conduct a type I and type II error analysis by using the observed effect size of ProThelial to determine adequacy of sample size of 66 patients using the P value _.001, powered at 95% confidence of no false negatives.
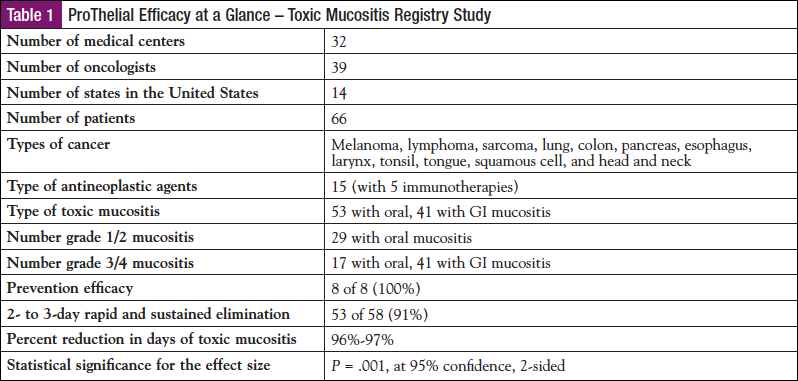
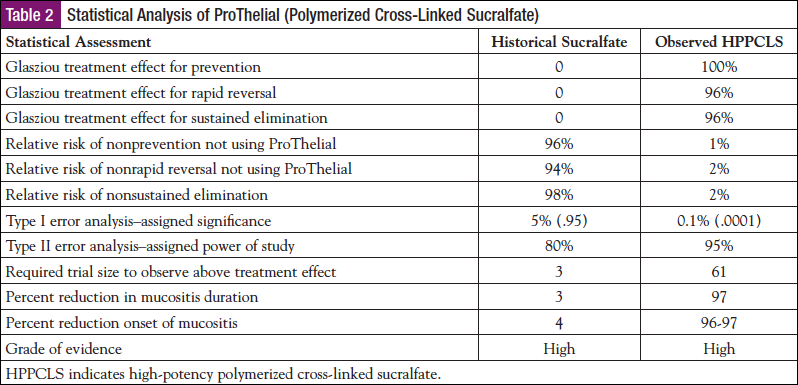
Results: The Glasziou treatment effect for prevention, rapid reversal, and sustained elimination was 100%, 96%, and 96%, respectively, for ProThelial versus 0% for generic sucralfate and MASCC-supported interventions. The type I and type II error with the ProThelial treatment effect at 0.1%, powered at 95%, predicted that the trial sample size required to verify a ProThelial treatment effect that reduces 70- to 84-day duration to 2 to 3 days was a total of 3 patients. The ProThelial trial had 61 patients with these outcomes versus the 3 that were statistically required. The 70- to 84-day duration of mucositis reduced to 2 to 3 days by ProThelial was a 96% to 97% reduction in mucositis duration, which is unprecedented. Complete prevention of toxic mucositis by ProThelial was equally unprecedented.
Conclusions:
- Complete prevention and/or rapid and sustained elimination of toxic mucositis
- Should reduce mucositis-associated costs
- Should eliminate mucositis-associated morbidity
- Should eliminate mucositis-associated mortality (an expected 20% 5-year mortality rate)
- Statistically significant evidence shows that ProThelial is associated with complete prevention and rapid sustained elimination of toxic mucositis during chemoradiation.
- ProThelial should be used in most patients receiving chemoradiation or undergoing HSCT.
References
- McCullough RW. A 66 patient multi-institution phase IV post-authorization surveillance of ProThelial (high potency polymerized cross-linked sucralfate) single agent efficacy for the prevention and rapid reversal of chemo-radiation induced oral, esophageal and intestinal mucositis. Brit J Med Medical Res. 2015;10:1-17.
- Elting LS, Keefe DM, Sonis ST, et al. Patient-reported measurements of oral mucositis in head and neck cancer patients treated with radiotherapy with or without chemotherapy: demonstration of increased frequency, severity, resistance to palliation, and impact on quality of life. Cancer. 2008;113:2704-2713.
- Cox JD, Stetz J, Pajak TF. Toxicity criteria of the Radiation Therapy Oncology Group (RTOG) and the European Organization for Research and Treatment of Cancer (EORTC). Int J Radiat Oncol Biol Phys. 1995;31:1341-1346.
- Franco P, Martini S, Di Muzio J, et al. Prospective assessment of oral mucositis and its impact on quality of life and patient-reported outcomes during radiotherapy for head and neck cancer. Med Oncol. 2017;34:81.
- Glasziou P, Chalmers I, Rawlins M, et al. When are randomised trials unnecessary? Picking signal from noise. BMJ. 2007;334:349-351.




