Background: Psychological distress (PD) is a known concern for many cancer survivors, and can be affected by factors such as illness perception (IP) and fear of cancer recurrence (FCR). Previous studies have addressed these variables independently among various groups of cancer survivors. Although survivors of gynecologic cancer account for approximately 9% of all cancer survivors, little is known about the relationship among these variables in this subpopulation.
Objectives: To explore the relationship between PD, IP, and FCR among survivors of gynecologic cancer, and to test whether this relationship differs by age, race, survivorship status, or survivorship duration.
Methods: An exploratory descriptive approach was used, along with a web-based survey. Data analyses were based on descriptive statistics, Pearson’s correlation coefficients, t-test results, Mann-Whitney U test findings, and a stepwise forward and backward multiple-regression model.
Results: Levels of PD, FCR, and IP were higher among early survivors and those who were young. PD, FCR, and IP were slightly lower for nonwhites than whites. IP and survivorship duration were the best predictors for PD among white participants. IP alone was the best predictor for PD among nonwhites.
Conclusions: FCR and IP play a role in the PD experienced by survivors of gynecologic cancer. Although IP has been shown to correlate with PD among survivors of other cancers, this study shows that IP also has a role in the PD of gynecologic cancer survivors. Although nonwhites experienced PD differently from whites, this finding should be interpreted with caution because of the small number of nonwhite participants.
Medical advancements, including early detection of cancer and the availability of effective treatments, have resulted in more patients living beyond their cancer diagnosis.1 Approximately 64% of adults diagnosed with cancer will be alive 5 years after their diagnosis.2 However, the overall increase in survival rates for patients with cancer has led to distressing symptoms, including anxiety, depression, and cognitive impairment, which may interfere with patients’ ability to perform daily activities.3 Although most cancer survivors adapt to their lives after cancer treatment, it has been recognized that, even for survivors who report excellent emotional adjustment, there are almost always areas of continuing disruption during the survivorship phase.4
Survivors of gynecologic cancer account for approximately 9% of all cancer survivors, and approximately 40% of patients in this subgroup reportedly have experienced some form of psychological distress (PD).5 In a cross-sectional study of 45 survivors of gynecologic cancer, 28.9% experienced clinical anxiety, 20.0% had mild-to-severe depression, and 15.6% had probable posttraumatic stress disorder.6 Younger survivors of endometrial cancer had greater clinical levels of anxiety, depression, and fear of recurrence.7 In a study of 49 survivors of ovarian cancer, 20% were considered “emotionally at-risk,” 22% expressed fear of recurrence, 36% indicated fear of developing a second cancer, and 6% were clinically depressed.8 In another study, nearly 28% of cervical cancer survivors met the criteria for clinical depression.9 Although a consensus has not been achieved, most research on the psychological impact of cancer survivorship has shown that this subpopulation of survivors experience residual sequelae of cancer, including PD.7-9 Fear of cancer recurrence (FCR) has been found to cause a significant emotional burden among patients with cancer, disrupting the psychological aspects of their life.8-10 Greater perceived risk for cancer recurrence has been associated with increased cancer worry among survivors of gynecologic cancer.5 An investigation conducted among long-term (>5 years) early-stage survivors of ovarian cancer showed that a significant amount of distress was related to the possibility of cancer recurrence (22%) and to fear of a second cancer (36%).8 Several studies indicate that FCR is a chief concern among survivors of gynecologic cancer; its prevalence ranges from 22% to 56%.8,11,12
In response to the high prevalence of FCR, the Institute of Medicine (IOM) has prioritized helping survivors understand and manage their fears.13 Although FCR is a known concern for many cancer survivors that can interfere with their quality of life (QOL) and general well-being,8,9,12 it is unclear how FCR relates specifically to the PD experienced by gynecologic cancer survivors.
Illness perception (IP) is another major determinant of QOL,14 and one’s own evaluation of the meaning of the cancer experience has emotional and behavioral consequences during cancer survivorship.15 Among men treated for localized prostate cancer, severely perceived consequences of prostate cancer were associated with poorer emotional well-being.16 In a study of 484 survivors of esophageal cancer, regression models indicated that the variables measured (perceptions and coping strategies) could explain 51% of the variance in anxiety and 42% of the variance in depression, most of which related to perceptions about esophageal cancer.17 An examination of the association between FCR, IP, medication beliefs, and side effects of treatment among women receiving adjuvant endocrine therapy after the diagnosis of breast cancer indicated that all aspects of IPs (apart from personal control) were associated with FCR.18 Results of studies involving patients treated for prostate cancer16 or esophageal cancer17 suggest that posttreatment emotional well-being can be influenced by cancer beliefs and expectations. Studies of IP among patients with illness of various types have provided empiric support that patients’ theories of illness are critical to successful adaptation to medical illness. However, a search of the medical literature did not reveal any studies of IP conducted among survivors of gynecologic cancer.
Research supports the fact that PD and FCR exist in cancer survivors, including those who had been diagnosed with gynecologic cancer. Although IP has been studied among survivors of various types of cancer, sufficient information on IP does not exist for survivors of gynecologic cancer. The IOM has urged healthcare providers to address the psychological needs of patients with cancer through the effective detection of distress and the provision of support services.19 Even though survivors of gynecologic cancer account for 9% of cancer survivors, little is known about the relationship between IP, FCR, and PD in this subpopulation, or whether such relationship differs by age, race, survivorship status, and/or survivorship duration. Better understanding of PD and related variables can assist in early recognition of the need for support services and in provision of appropriate services. The purpose of this study was to explore the relationship between PD, IP, and FCR among survivors of gynecologic cancer.
Aims of the study were (1) to explore the characteristics of PD, IP, and FCR across age-groups, survivorship status, and racial groups (whites vs nonwhites), and (2) to explore the relationships between PD, IP, and FCR with age and survivorship duration within each racial group. The following research questions were used to address the study aims:
- What are the distribution characteristics of PD, IP, and FCR among age-groups, survivorship status, and racial groups?
- What is the relationship between PD, FCR, and IP to age and survivorship duration within racial groups?
- What are the differences in PD, IP, and FCR across age-groups, survivorship status, and racial groups?
- Does IP, FCR, age, or survivorship duration predict PD for white or nonwhite survivors of gynecologic cancer?
Design
Methodology
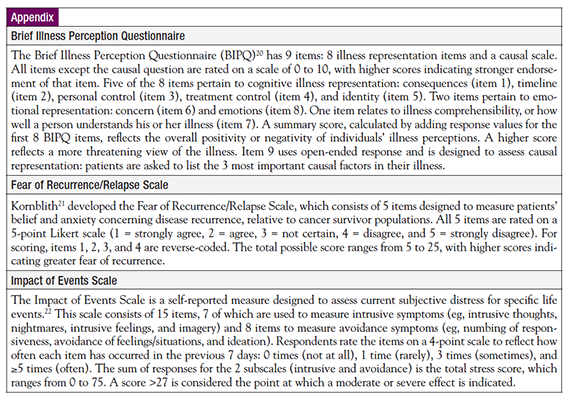
This study was approved by the Institutional Review Board of The University of Texas Medical Branch at Galveston. An exploratory, cross-sectional, descriptive research approach, using a web-based survey, was used to collect data. A survey link was created in SurveyMonkey, and 4 questionnaires were uploaded, including a demographic questionnaire, the Brief Illness Perceptions Questionnaire (BIPQ),20 Fear of Recurrence/Relapse (FOR) Scale,21 and the Impact of Events Scale (IES22; see Appendix). Participants were invited by means of recruitment flyers that were placed at local clinical facilities, announcements through online cancer support groups, and a Facebook page that was managed by the author, who served as principal investigator for the study. Participation was voluntary. To preserve anonymity, written consent was waived. Data were collected from March 23, 2013, through May 17, 2013.
The transition theory of Meleis23 was used as the conceptual framework for this study, because cancer survivorship is considered a transition from the diagnosis stage. The 3 concepts from this framework included (1) type of transition, (2) transition conditions, and (3) patterns of response or outcome response. Cancer survivorship was considered a “health and illness” type of transition. Transition conditions included demographic factors, diagnosis, disease-free interval since treatment completion, IP, and FCR. The pattern of response or outcome in this study was PD. Other concepts are beyond the scope of this study.
Sample
The study sample comprised women aged ≥21 years with a history of uterine/endometrial, ovarian, or cervical cancer who had been disease-free for ≥2 years since completion of cancer treatment. Excluded from participation were patients aged <21 years, those unable to read and write in English, and those with a history of vulvar, vaginal, or nongynecologic cancer.
Research Variables and Instruments
The study variables were IP, FCR, and PD. IP was defined as the beliefs held by patients about their health problems.24 IP was assessed using Broadbent and colleagues’ BIPQ.20 FCR was defined as the experience of worry and concern that cancer may return to the same organ or spread to another part of the body.25 FCR was ascertained with Kornblith’s FOR Scale.21 PD involves an inability to cope, emotional changes, discomfort communicated to medical professionals, and harm.26 PD warrants treatment by a nursing staff, and is a more appropriate term to describe a patient’s response to illness than “distress” alone.26 PD also has been defined as a concept of “maladaptive psychological functioning in the face of stressful life events.”27 PD was evaluated with the IES, which was developed by Horowitz and colleagues.22 The demographic variables included age, race, diagnosis, and disease-free interval since treatment completion. In this study, cancer survivors were defined as those who completed treatment ≥2 years before data collection and remained disease-free through study participation. Patients who were disease-free for 2 to 5 years since treatment completion were considered “early survivors,” and those with >5 disease-free years since the conclusion of treatment were considered “long-term survivors.” For this study, survivorship was measured using survivorship status (early vs long-term survivors) and survivorship duration (number of disease-free years).
Data Analyses
Data analyses were based on descriptive statistics, Pearson’s correlation coefficients, t-test results, Mann-Whitney test findings, and a stepwise forward and backward multiple-regression model. Data were analyzed using SPSS software (version 21.0; IBM, Armonk, NY), and significance was defined as α = .05. All data were examined for normality and homogeneity.
Results
Of the 632 women who responded to the survey, 376 were eligible to participate in the study. Of the 376 eligible participants, 352 completed all 4 questionnaires, and their responses were used in the final analyses. All instruments demonstrated respectable reliability: BIPQ, α = .753; FOR Scale, α = .845; and IES, α = .892. The mean age of participants was 57.07 years (standard deviation [SD], 10.12; range, 29-80 years). The mean disease-free interval (survivorship duration) was 5.65 years (SD, 4.71; range, 2-30 years). Women who responded to the survey represented 5 races/ethnicities, including non-Hispanic white (88.9%), Hispanic white (3.7%), African American or black (1.4%), Hispanic (2.3%), and Asian (3.7%). The majority of participants (59.7%) were survivors of ovarian cancer, 31.2% were survivors of uterine or endometrial cancer, and 9.1% were survivors of cervical cancer.
Excluding non-Hispanic whites, ethnic groups were combined into a “nonwhite” racial group for subsequent analyses. Whites represented 92.6% of the study population, and nonwhites represented 7.4%. Two age-groups were created using the mean split (age 57 years): age 29 to 57 years (47.2%) and age 58 to 80 years (52.8%). Survivorship was measured as an interval variable based on the number of disease-free years (survivorship duration) and as a dichotomous variable (survivorship status: early [2-5 years] vs long-term [>5 years] survival since treatment completion). There were more early survivors (65.6%) than long-term survivors (34.4%). The mean scores for PD, FCR, and IP were lower for nonwhites, indicating that these participants experienced less PD, FCR, and IP than white participants (Table 1).
The older age-group had lower mean scores for PD and IP than the younger age-group, indicating that the older survivors experienced less PD and IP. The FCR score was slightly higher for the younger age-group, reflecting slightly higher FCR among these survivors (Table 2). Mean scores for PD, FCR, and IP were higher for early survivors than long-term survivors, indicating that shorter time since treatment completion was associated with greater PD, FCR, and IP (Table 3).
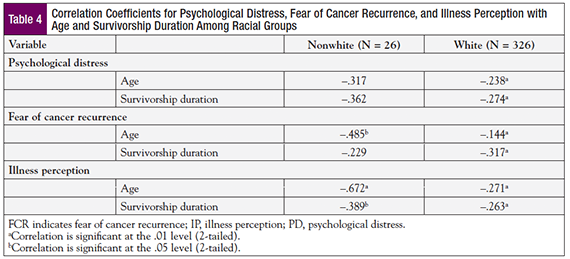
An overall pattern of negative correlation between PD, FCR, and IP with age and survivorship duration was noted for whites and nonwhites; this indicates that as age and survivorship duration increased, PD, FCR, and IP decreased (Table 4). Age had a moderate or strong correlation with all 3 study variables for nonwhites, but its relationship with PD was not significant, perhaps because of the small sample size. Conversely, for whites, the relationship between age and all 3 study variables was significant, but the effect sizes were extremely small. The correlations between survivorship duration and PD and IP were stronger for nonwhites than whites.
Further evaluation was performed to examine the differences between groups. The age-group differences in PD, FCR, and IP were evaluated using t tests (Table 5). The differences in PD, FCR, and IP were significant between the younger and older age-groups, suggesting that the younger participants had experienced more PD, FCR, and IP than the older participants.
As a result of sample-size differences in the racial and survivorship groups, as well as the small samples of nonwhites and long-term survivors, a nonparametric statistical test (Mann-Whitney) was used to examine the differences in PD, IP, and FCR between racial groups and survivorship status. No significant difference in PD or IP was observed between whites and nonwhites (Table 6); however, for FCR, a marginally significant difference was found. The FCR mean rank was lower for nonwhites than whites (P = .065). Overall, these findings appear to suggest that race did not have a significant impact on PD, FCR or IP. Results of the Mann-Whitney test of the difference between early and long-term survivors (Table 7) showed significant differences in PD, FCR, and IP, indicating that early survivors experienced more PD, greater fear of recurrence, and more consequences from their illness than long-term survivors (P <.001).
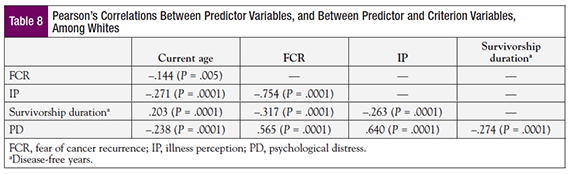
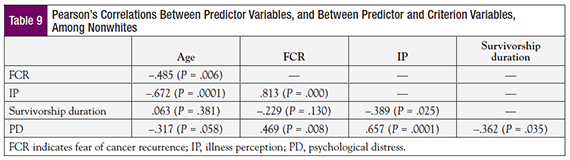
A Pearson’s correlation coefficient was calculated to determine the correlation between predictors, and between predictors and the criterion, among white participants. FCR and IP were found to correlate strongly (r = .754, P = .0001; Table 8). Although correlations were found for other predictive variables, their magnitude was small or moderate. A Pearson’s correlation coefficient also was calculated for nonwhites (Table 9), which showed that the correlation between FCR and IP was even stronger for this group (r = .813, P = .0001). With respect to age, strong negative correlations were observed for IP (r = –.672, P = .0001) and FCR (r = –.485, P = .006). A significant negative correlation was observed for IP and survivorship duration (r = –.389, P = .025).
A stepwise forward and backward multiple-regression analysis was conducted to evaluate whether IP, FCR, age, race, or survivorship duration predicted PD among survivors of gynecologic cancer (Table 10). IP and survivorship were the best predictors for PD among white participants, and IP alone was the best predictor for PD among nonwhites.
Discussion
Previous studies have shown that PD and FCR exist in cancer survivors, and these variables have been studied independently for other types of cancer.28,29 In a study of 321 older survivors (aged >60 years) of breast, colorectal, or prostate cancer, PD was lower among African Americans than whites.28 Applying data from the National Health Interview Survey (2005-2007), Shootman and colleagues found that among survivors of various types of cancer, including bladder, breast, cervical, uterine, ovarian, colorectal, lung, lymphoma, prostate, and thyroid cancer, PD was more prevalent among whites than African Americans.29 The current study also demonstrated that white participants with gynecologic cancer reported greater PD than nonwhite participants with gynecologic cancer. However, a previous study of long-term survivors of adult-onset cancer showed no correlation between race and serious PD.1
The present study demonstrated that PD appears to be lower among older participants. This result is consistent with findings from a study of survivors of breast and endometrial cancer, in which younger age was found to predict higher distress.7 In the current study, survivorship duration (number of disease-free years) correlated significantly with PD, FCR, and IP among whites. However, among nonwhites, only IP correlated significantly with survivorship duration. This study also showed a correlation between disease-free years and PD. In contrast, a study of PD in long-term survivors of adult-onset cancer (≥5 years) showed no association between PD and the number of years since cancer diagnosis.1 Similarly, an examination of psychological outcomes of long-term survival after gynecologic cancer showed no relationship between distress and the number of years since diagnosis.12 The difference in findings among these studies may be attributable to differences in sample sizes.
It appears that no published study had examined IP specifically among survivors of gynecologic cancer, but the literature does support that a person’s evaluation of the meaning of the cancer experience has emotional and behavioral consequences during cancer survivorship.15 Similar to the current study, an evaluation of IP among patients with thyroid cancer demonstrated a negative correlation between IP and time since treatment, indicating that as survivorship duration increases, IP decreases.14 However, the correlation between IP and survivorship duration in the current study was significant only for nonwhites. Moreover, older participants in the present study had fewer IPs. In contrast, the study of patients with thyroid cancer showed no correlation between IP and age.14 The differences in findings may be attributable to the type of cancer. A study of women with breast cancer who underwent adjuvant endocrine therapy demonstrated that all aspects of IP (except personal control) correlated significantly with FCR.18 The present study showed that, among survivors of gynecologic cancer, nonwhite participants perceived fewer consequences resulting from their cancer; this finding is consistent with those of studies that addressed ethnic differences in IP among diabetic patients30 and patients undergoing maintenance hemodialysis.31 Previous studies have shown correlations between IP and PD16,32 and between IP and FCR18 among patients with cancer or survivors of cancer. For survivors of esophageal cancer, IP explained the majority of variance in anxiety and depression.32
In another study, FCR was found to correlate with high levels of anxiety and depression among patients with pancreatic cancer who were examined after potentially curative pancreatectomy.33 Data for breast cancer survivors indicate that younger age was consistently associated with higher FCR, suggesting that FCR differs by age,34 which is consistent with findings of the present study.
Although the present study showed a marginally significant difference in FCR between whites and nonwhites, a prospective study of patients with breast cancer did not demonstrate differences in FCR between races.34 In a study of IP across ethnicities that involved patients with diabetes (86 Europeans, 86 Asians, and 87 Pacific Islanders), Bean and colleagues found that scores were higher for Pacific Islanders for 3 subscales of the BIPQ: diabetes consequences, identity, and emotional representations.30
Both similarities and differences were found in the relationships of PD, FCR, and IP with age and survivorship duration between this study and other research. These differences may relate to differences in cancer type and race/ethnicity. Moreover, sufficient previous data are lacking for IP among survivors of gynecologic cancer, which hinders generalizations and comparisons.
Implications for Research and Practice
As more advanced practice nurses participate in the care of cancer survivors and patients with cancer, a better understanding of PD and related variables can assist in the development of interventions to address the PD experienced by gynecologic cancer survivors. Because the evaluation of IP and its relation to PD among these survivors has been limited, a cross-study comparison is not yet possible. It is recommended that future research of gynecologic cancer survivors includes direct data collection rather than online surveys to control participation based on age, diagnosis, and race. Although direct data collection may be more costly and time-consuming, it may improve the generalizability of the findings. Nurses play a vital role in PD screening among patients with cancer and survivors of cancer, and require knowledge and skill to identify PD and refer patients appropriately. Therefore, it is important to incorporate these aspects into training so that nurses can better assist cancer survivors who already have PD and effectively screen patients who are at risk for PD.
Limitations
Limitations include overrepresentation of ovarian cancer survivors and white women. Moreover, study participation was limited to those who speak English. These characteristics limit the generalizability of the results to nonwhite and non–English-speaking cancer survivors. Less common gynecologic cancers (eg, vulvar, vaginal) were not included in this study; therefore, results may not be applicable to survivors of less common gynecologic malignancies. Other limitations include self-selection bias, self-reporting bias, and potential inaccuracies in self-identified characteristics. This study entailed a cross-sectional survey and, thus, did not allow for any conclusions to be drawn about changes in PD over time.
Conclusions
The overall conclusion is that survivors of gynecologic cancer experience PD, and that FCR and IP play a role in the PD experienced by these survivors (Table 11). These findings align closely with those of researchers who have studied these concepts in patients with other types of cancers. Although the present study indicates that nonwhites may experience PD differently from whites, this finding should be interpreted with caution because of the small number of nonwhite participants. Moreover, it may be concluded that younger cancer survivors and early survivors experience more PD, FCR, and IP than older and long-term survivors. The findings also indicate that PD, FCR, and IP decrease as the disease-free interval increases. The study results suggest that survivors of gynecologic cancer experience PD and, consequently, these patients should be screened and referred to the appropriate services.
References
- Hoffman KE, McCarthy EP, Recklitis CJ, Ng AK. Psychological distress in long-term survivors of adult-onset cancer: results from a national survey. Arch Intern Med. 2009;169:1274-1281.
- Siegel R, DeSantis C, Virgo K, et al. Cancer treatment and survivorship statistics, 2012. CA Cancer J Clin. 2012;62:220-241.
- Zabora J, BrintzenhofeSzoc K, Curbow B, et al. The prevalence of psychological distress by cancer site. Psychooncology. 2001;10:19-28.
- Meyerowitz BE, Kurita K, D’Orazio LM. The psychological and emotional fallout of cancer and its treatment. Cancer J. 2008;14:410-413.
- Matulonis UA, Kornblith A, Lee H, et al. Long-term adjustment of early-stage ovarian cancer survivors. Int J Gynecol Cancer. 2008;18:1183-1193.
- Urbaniec OA, Collins K, Denson LA, Whitford HS. Gynecologic cancer survivors: assessment of psychological distress and unmet supportive care needs. J Psychosoc Oncol. 2011;29:534-551.
- Kornblith AB, Powell M, Regan MM, et al. Long-term psychosocial adjustment of older vs younger survivors of breast and endometrial cancer. Psychooncology. 2007;16:895-903.
- Wenzel LB, Donnelly JP, Fowler JM, et al. Resilience, reflection, and residual stress in ovarian cancer survivorship: a gynecologic oncology group study. Psychooncology. 2002;11:142-153.
- Bradley S, Rose S, Lutgendorf S, et al. Quality of life and mental health in cervical and endometrial cancer survivors. Gynecol Oncol. 2006;100:479-486.
- Cimprich B, Ronis DL, Martinez-Ramos G. Age at diagnosis and quality of life in breast cancer survivors. Cancer Pract. 2002;10:85-93.
- Dahl L, Wittrup I, Væggemose U, et al. Life after gynecologic cancer--a review of patients quality of life, needs, and preferences in regard to follow-up. Int J Gynecol Cancer. 2013;23:227-234.
- Hodgkinson K, Butow P, Fuchs A, et al. Long-term survival from gynecologic cancer: psychosocial outcomes, supportive care needs and positive outcomes. Gynecol Oncol. 2007;104:381-389.
- Hewitt M, Greenfield S, Stovall E. From Cancer Patient to Cancer Survivor: Lost in Transition. Washington, DC: National Academies Press; 2006.
- Hirsch D, Ginat M, Levy S, et al. Illness perception in patients with differentiated epithelial cell thyroid cancer. Thyroid. 2009;19:459-465.
- Bowman KF, Deimling GT, Smerglia V, et al. Appraisal of the cancer experience by older long-term survivors. Psychooncology. 2003;12:226-238.
- Traeger L, Penedo FJ, Gonzalez JS, et al. Illness perceptions and emotional well-being in men treated for localized prostate cancer. J Psychosom Res. 2009;67:389-397.
- Dempster M, McCorry NK, Brennan E, et al. Psychological distress among survivors of esophageal cancer: the role of illness cognitions and coping. Dis Esophagus. 2012;25:222-227.
- Corter AL, Findlay M, Broom R, et al. Beliefs about medicine and illness are associated with fear of cancer recurrence in women taking adjuvant endocrine therapy for breast cancer. Br J Health Psychol. 2013;18:168-181.
- Adler NE, Page AEK. Institute of Medicine (US) Committee on Psychosocial Services to Cancer Patients/Families in a Community Setting. Washington, DC: National Academies Press; 2008.
- Broadbent E, Petrie KJ, Main J, Weinman J. The Brief Illness Perception Questionnaire. J Psychosom Res. 2006;60:631-637.
- Kornblith AB. Psychosocial adaptation of cancer survivors. In: Holland J, ed. Psychooncology. New York, NY: Oxford University Press; 1998:223-254.
- Horowitz M, Wilner N, Alvarez W. Impact of Event Scale: A measure of subjective stress. Psychosom Med. 1979;41:209-218.
- Meleis AI. Transition theory. In: Tomey AM, Alligood MR, eds. Nursing Theorists and Their Work. 7th ed. St Louis, MO: Mosby Elsevier; 2000:416-433.
- Petrie KJ, Broadbent E, Kydd R. Illness perceptions in mental health: issues and potential applications. J Ment Health. 2008;17:559-564.
- Vickberg SM. The Concerns About Recurrence Scale (CARS): a systematic measure of women’s fears about the possibility of breast cancer recurrence. Ann Behav Med. 2003;25:16-24.
- Ridner SH. Psychological distress: concept analysis. J Adv Nurs. 2004;45:536-545.
- Abeloff MD, Armitage JO, Lichter AS, Niederhuber JE. Clinical Oncology. 2nd ed. New York, NY: Churchill Livingstone; 2000.
- Deimling GT, Wagner LJ, Bowman KF, et al. Coping among older adult, long-term cancer survivors. Psychooncology. 2006;15:143-159.
- Schootman M, Deshpande AD, Pruitt SL, et al. National estimates of racial disparities in health status and behavioral risk factors among long-term cancer survivors and non-cancer controls. Cancer Causes Control. 2010;21:1387-1395.
- Bean D, Cundy T, Petrie KJ. Ethnic differences in illness perceptions, self-efficacy and diabetes self-care. Psychol Health. 2007;22:787-811.
- Kim Y, Pavlish C, Evangelista LS, et al. Racial/ethnic differences in illness perceptions in minority patients undergoing maintenance hemodialysis. Nephrol Nurs J. 2012;39:39-49.
- Dempster M, McCorry NK, Brennan E, et al. Illness perceptions among carer–survivor dyads are related to psychological distress among oesophageal cancer survivors. J Psychosom Res. 2011;70:432-439.
- Petzel MQ, Parker NH, Valentine AD, et al. Fear of cancer recurrence after curative pancreatectomy: a cross-sectional study in survivors of pancreatic and periampullary tumors. Ann Surg Oncol. 2012;19:4078-4084.
- Liu Y, Pérez M, Schootman M, et al. Correlates of fear of cancer recurrence in women with ductal carcinoma in situ and early invasive breast cancer. Breast Cancer Res Treat. 2011;130:165-173.